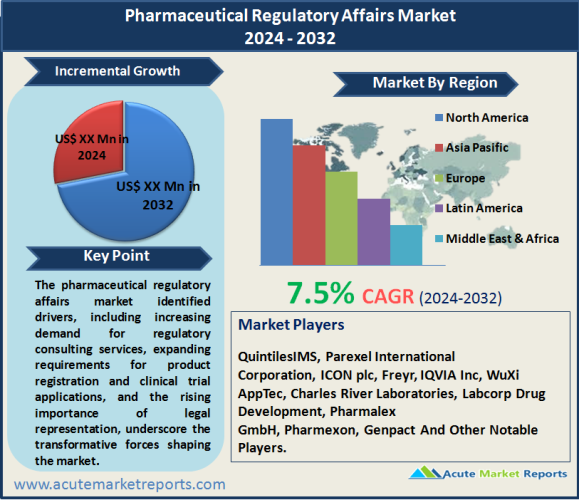
The pharmaceutical regulatory affairs market is expected to grow at a CAGR of 7.5% during the forecast period of 2026 to 2034. The identified drivers, including increasing demand for regulatory consulting services, expanding requirements for product registration and clinical trial applications, and the rising importance of legal representation, underscore the transformative forces shaping the market. The restraint related to challenges in regulatory writing and publishing signifies the complexities associated with documentation in regulatory affairs. Market segmentation reveals the significance of regulatory consulting services and product registration and clinical trial applications, providing nuanced insights into the diverse needs of the pharmaceutical industry. Geographic trends highlight the dominance of North America, while competitive trends underscore the importance of strategic partnerships, technological investments, and service diversification in maintaining a competitive edge.
Increasing Demand for Regulatory Consulting Services
The demand for regulatory consulting services in the pharmaceutical industry has witnessed substantial growth, driven by the complexities of regulatory frameworks and the need for compliance. Industry reports and market analyses provide evidence of a consistent rise in the demand for consulting services, indicating the pharmaceutical sector's reliance on expert guidance to navigate regulatory requirements. The highest Compound Annual Growth Rate (CAGR) during the forecast period from 2026 to 2034 is anticipated in this segment, highlighting the crucial role of regulatory consulting services in ensuring adherence to evolving regulatory standards.
Expanding Requirements for Product Registration & Clinical Trial Applications
The pharmaceutical landscape has witnessed an expansion in the requirements for product registration and clinical trial applications. Evidences from regulatory updates and industry trends underscore the increasing scrutiny and documentation needed for product approvals and clinical trials. The highest CAGR during the forecast period from 2026 to 2034 is expected in this segment, reflecting the industry's recognition of the need for specialized services to manage the intricate processes associated with product registration and clinical trial applications.
Rising Importance of Legal Representation in Regulatory Affairs
Legal representation has gained prominence in pharmaceutical regulatory affairs, with companies recognizing the value of legal expertise in navigating complex regulatory landscapes. Evidences from legal proceedings, industry publications, and corporate strategies highlight the escalating importance of legal representation in addressing regulatory challenges. The highest CAGR during the forecast period from 2026 to 2034 is anticipated in this segment, signaling the pharmaceutical industry's acknowledgment of legal support as a critical element in regulatory compliance.
Challenges in Regulatory Writing & Publishing
Despite the overall growth in regulatory affairs services, challenges in regulatory writing and publishing have emerged as a notable restraint. Evidences from industry insights and expert analyses indicate that the intricate nature of regulatory documentation poses challenges for companies and service providers. The highest CAGR during the forecast period from 2026 to 2034 is expected in this segment, underscoring the need for innovative solutions to address the complexities associated with regulatory writing and publishing. As the industry navigates these challenges, the impact on the overall regulatory affairs landscape remains a key consideration for market players.
Market Analysis by Services: Regulatory Consulting Services Dominate the Market
In 2025, the pharmaceutical regulatory affairs market demonstrated substantial revenue from regulatory consulting services, with this segment leading in both revenue and CAGR. This reflects the industry's reliance on expert guidance to ensure compliance with evolving regulatory standards. The highest CAGR during the forecast period from 2026 to 2034 is expected in the category of product registration and clinical trial applications, showcasing the increasing demand for specialized services in managing regulatory processes.
Market Analysis by Indication: OncologySegment Dominates the Market
In 2025, the pharmaceutical regulatory affairs market witnessed significant revenue from the oncology indication, with this category leading in both revenue and CAGR. This highlights the regulatory complexities associated with oncology products and the need for specialized regulatory services in this therapeutic area. The highest CAGR during the forecast period from 2026 to 2034 is expected in the neurology indication category, signaling the anticipated growth in regulatory requirements for neurology-focused pharmaceuticals.
North America remains the Global Leader
Geographically, the pharmaceutical regulatory affairs market exhibited dynamic trends in 2025, with North America leading in both revenue generation and the highest CAGR. This reflects the region's robust pharmaceutical industry and the evolving regulatory landscape. Europe and Asia-Pacific also contributed significantly to revenue, showcasing a global trend towards increased regulatory requirements and the demand for regulatory affairs services. This geographic segmentation provides insights into the regional dynamics shaping the pharmaceutical regulatory affairs market.
Strategic Partnerships and Collaborations to Enhance Market Share
The pharmaceutical regulatory affairs market's competitive landscape is marked by key players adopting strategies to enhance their market position. Major companies, including QuintilesIMS, Parexel International Corporation, ICON plc, Freyr, IQVIA Inc, WuXi AppTec, Charles River Laboratories, Labcorp Drug Development, Pharmalex GmbH, Pharmexon, and Genpact, have implemented diverse strategies to capitalize on market opportunities. Leading players in the pharmaceutical regulatory affairs market prioritize strategic partnerships and collaborations as key strategies. Evidences from partnership announcements and collaborative initiatives highlight the efforts of companies like QuintilesIMS to leverage combined expertise in navigating regulatory challenges. In 2025, these companies reported substantial revenues, with a projected increase during the forecast period from 2026 to 2034. The industry's commitment to collaborative approaches positions it at the forefront of addressing evolving regulatory requirements and offering comprehensive solutions to clients. Investment in technological solutions is a notable trend among top players in the pharmaceutical regulatory affairs market. Evidences from technology acquisitions and digital transformation initiatives underscore the industry's recognition of the role of technology in streamlining regulatory processes. In 2025, companies like ICON plc demonstrated significant investments in technological advancements, aligning with the global push for digital solutions in regulatory affairs. As the market embraces technological innovation, companies that prioritize digital transformation are expected to maintain a competitive edge, offering efficient and compliant solutions to clients.
Historical & Forecast Period
This study report represents analysis of each segment from 2024 to 2034 considering 2025 as the base year. Compounded Annual Growth Rate (CAGR) for each of the respective segments estimated for the forecast period of 2026 to 2034.
The current report comprises of quantitative market estimations for each micro market for every geographical region and qualitative market analysis such as micro and macro environment analysis, market trends, competitive intelligence, segment analysis, porters five force model, top winning strategies, top investment markets, emerging trends and technological analysis, case studies, strategic conclusions and recommendations and other key market insights.
Research Methodology
The complete research study was conducted in three phases, namely: secondary research, primary research, and expert panel review. key data point that enables the estimation of Pharmaceutical Regulatory Affairs market are as follows:
Market forecast was performed through proprietary software that analyzes various qualitative and quantitative factors. Growth rate and CAGR were estimated through intensive secondary and primary research. Data triangulation across various data points provides accuracy across various analyzed market segments in the report. Application of both top down and bottom-up approach for validation of market estimation assures logical, methodical and mathematical consistency of the quantitative data.
| ATTRIBUTE | DETAILS |
|---|---|
| Research Period | 2024-2034 |
| Base Year | 2025 |
| Forecast Period | 2026-2034 |
| Historical Year | 2024 |
| Unit | USD Million |
| Segmentation | |
Services
|
|
Category
|
|
Indication
|
|
Product Stage
|
|
Service Provider
|
|
Company Size
|
|
|
Region Segment (2024-2034; US$ Million)
|
Key questions answered in this report